[amazon_link asins=’B00ICSBNYE,B003PRGP22,B01M5E4XWF,B011AALC4Q,B01ASDGIJA’ template=’ProductCarousel’ store=’finmeacur-20′ marketplace=’US’ link_id=’b52ed34b-0540-11e7-aba3-0d1052d3ded9′]
Men at an above-normal risk of prostate cancer may be able to reduce their risk of developing the disease by taking a drug already on the market.
In research reported yesterday, the drug dutasteride, currently used to shrink enlarged prostates, was found to reduce the risk of prostate cancer by about a quarter in high-risk men.
The drug apparently caused small tumors to stop growing or even to shrink, a research team reported in the New England Journal of Medicine.
The medication is sold under the brand name Avodart.
A previous study had found that a similar drug, finasteride, could also lower the risk of prostate tumors, but the new research – conducted at 250 sites in 42 countries – suggests that dutasteride is slightly more effective.
The new study “is further evidence that there is a role for these drugs in risk reduction,” said Dr. Jack Jacoub, a medical oncologist at Orange Coast Memorial Medical Center in Fountain Valley, Calif., who was not involved in the study. “If a patient understands all the issues [associated with the drug], I think it would be appropriate to provide it.”
Dr. Howard M. Sandler, an oncologist at the Cedars-Sinai Medical Center in Los Angeles, was even more emphatic.
“The question might be, why isn’t every man taking one of these drugs?” he said. “They help people urinate better by shrinking the prostate, they probably reduce baldness, and they reduce the risk of prostate cancer. There seems to be very little downside to them.”
GlaxoSmithKline, which manufactures Avodart, said on Monday that it would apply to the Food and Drug Administration for permission to market the drug for risk reduction in men with high PSA levels, a measure of prostate cancer risk; a family history of the disease, or other risk factors such as ethnicity.
But because the drug is already available, doctors do not need to wait for such permission to prescribe it as a preventive.
Considering the low risk of the drug, that might be a safe option, experts said. Insurance companies are not likely to pay for it for that purpose, however, until the FDA approves it.
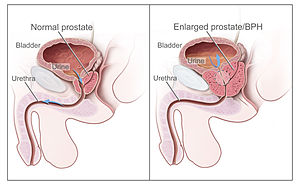
Both dutasteride and finasteride are already approved for treating benign prostatic hyperplasia, or BPH, an enlargement of the prostate gland that causes urinary and other problems.
Finasteride is sold by Merck & Co. Inc. under the brand name Proscar.
In the study, researchers enrolled 8,231 men, ages 50 to 75, who had elevated levels of PSA but no evidence of prostate tumors on a biopsy.
Half received dutasteride daily for four years; half received a placebo.
All the men received biopsies two years after enrollment and again two years later.
Overall, 659 men taking dutasteride were diagnosed with prostate cancer (19.9 percent), compared to 858 men (25.1 percent) taking a placebo – a 23 percent reduction.
Among men with a family history of prostate cancer, the drug reduced risk by 31.4 percent.
Price may be a problem. According to GlaxoSmithKline, the wholesale cost of Avodart is $3.23 per pill. Finasteride is available in a generic form and is thus cheaper.
Prostate cancer is the most common cancer in men after skin cancer. It affects 192,000 men a year and kills 27,000.
Source : The Blade : April ’01. 2010
![Reblog this post [with Zemanta]](https://i0.wp.com/img.zemanta.com/reblog_e.png?w=580)



